On the 15th November, the National Health and Medical Research Council (NHMRC) and Consumers Health Forum (CHF) hosted a 'workshop' to launch the review of the Statement on consumer and community involvement in health and medical research. Five speakers - Anne Mackenzie AM, Dr Jack Nunn, Dr Janelle Bowden, Dr Susan Ramos, and Debra Langridge - presented their views around the Statement and its topic, and the NHMRC/CHF asked some preliminary questions and hosted discussion on the topic.
The session was not able to be recorded. So for background reference and transparency, I (Janelle) am making the contents of my presentation available below.
This workshop was just the opening conversation for a review process that is yet to be defined, and will likely take more than a year to complete properly. We will continue to provide updates as we hear about them via the CCReW newsletter and the Research Gamechangers Facebook group for anyone interested in its progress and opportunities to input. You can also keep track of updates yourself, including a workshop summary and more, via the related NHMRC website or by signing up for the fortnightly NHMRC Tracker.
My reflections on consumer engagement and involvement
Good afternoon everyone and thank you to the NHMRC and CHF teams for the opportunity to share a few of my reflections today on consumer engagement and involvement in research.
I would like to begin by acknowledging that I’m presenting to you today from the land of the Wallumedegal clan in Sydney as well as recognise the unceded lands from which you are all listening to this webinar from today. I pay my respects to their elders past, present and emerging, as well as any first Nations peoples that may be joining us online today.
Over the next few minutes I’m going to provide some reflections on why I think consumer involvement and partnership matters, provide a bit of a refresher on when and how it impacts and some thoughts to a future statement for consideration.
So to cut to the chase we know that diversity strengthens teams and can lead to better project outcomes. Different eyes on the same problem can give you a new appreciation of a problem leading to new ideas for how to address it. In a team of researchers and clinician’s, consumers will often provide a different lens to the problem at hand.
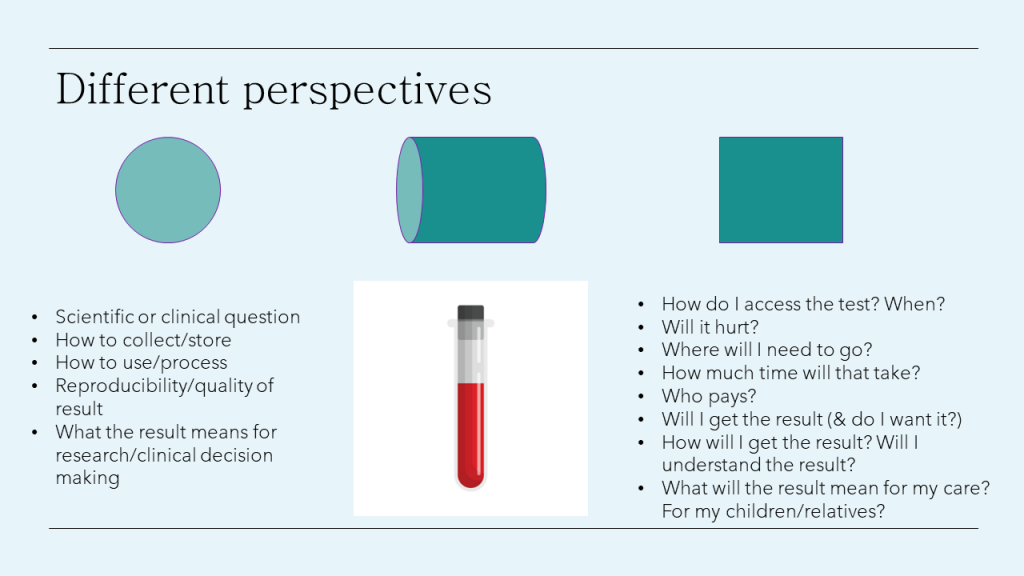
If we take for example research into a new biomarker which could lead to a new diagnostic.
From a researcher or clinician perspective you might be interested in a biomarker for its scientific or clinical value. In conducting the research, you might be thinking through - can we detect it? In order to detect it - how and when will we need to collect, store the sample (what tubes, temp), how we would process and use the samples (what methods, machines, people), how do we ensure the quality and the reproducibility that result? What will knowing that information or how to test for it means research or clinical decision-making?
Consumers on the other hand might have very different questions relating to access or the consequences of the knowing the result. How might the test be accessed - when, where, how much time will that take? What is required to do the test, and will it hurt, be invconvenient? Who will pay? Will I get a result (and do I actually want to know)? If yes, how, when and from whom will I get the result, and I will understand the result? What could the implications be for my care, and for their broader family. These practical questions might not seem that important to the researcher/clinical team members, but could have big implications to patients and hence, impact the translation and implementation of that research.
This is just one example but it really aims to demonstrate the value on having that practical experience of the condition and all the healthcare system within a research team.
So when, and where can people be involved in research?
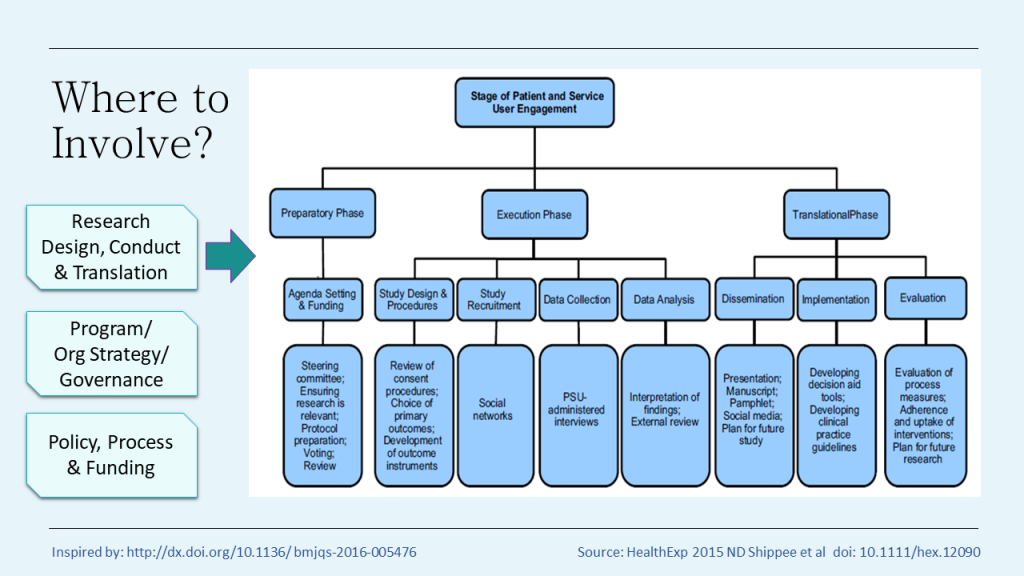
There are opportunities for involvement across the research cycle right from setting the agenda and priorities for research, to throughout the conduct of the research and beyond. There is no one way, but the earlier the involvement the greater potential to reap the benefits of that end user experience. Consumers should be involved from the very start in the prioritisation and design of research. But even when the study is over there is opportunity to broaden the relevance and reach of your research through partnering with consumers.
But it is not only in specific research projects where consumers can be involved. They can also contribute to program or organisational strategy and governance, in the policy and process space and helping decide what gets funded.
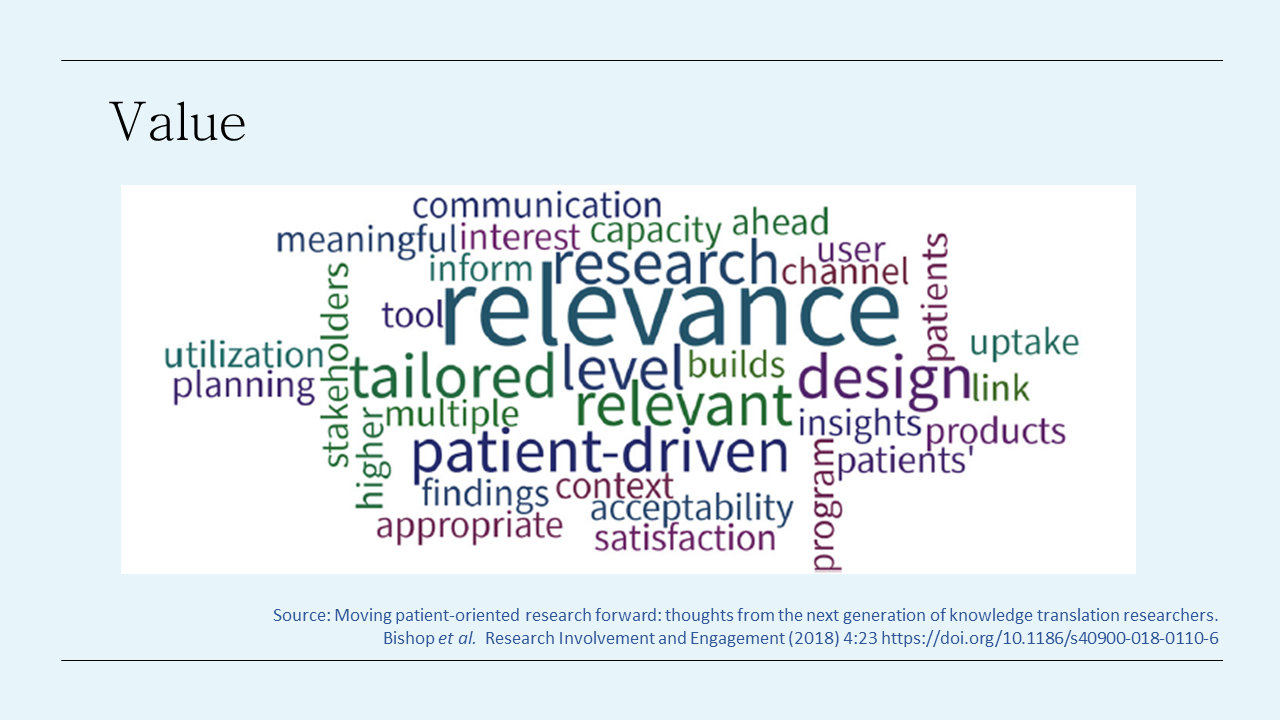
This word cloud is taken from a Canadian workshop with researchers a few years ago talking about the value of involving patients in research. I think it captures very nicely many of the benefits of consumers involvement – from improved relevance, acceptability, appropriateness, to ensuring research is patient-driven, improves satisfaction with the research and can help with communication.
And the impact can extend right across the research ecosystem and society at large. It can impact on what is researched, how it’s researched, the consent process. It can impact on the people involved, the research participants, the researchers and the wider community organisations, in the uptake and implementation of research and in driving changes in practice.

And that is simply because patients, the ultimate beneficiaries of medical and health research, empowered by the information available on the internet and their own patient communities, can often be more informed than clinicians about the healthcare, the practices, the research, the treatments that may have a positive impact on the quality and length of life, and will fight to make them available and accessible.
And to be more specific about how those impacts can be achieved using the example of a clinical trial involving consumers can answer questions like these...

Does the question matter to patients? Is there an unmet need? Is the research grounded in reality? are the right comparators in use, Are the eligibility criteria and protocol requirement practical? Do these patients exist? Will they accept the risk of what you’re proposing? Can they comply with, the tests, the visit schedule. Are we measuring outcomes that matter to Patients?
How will people find out about the trial? Consumers often have networks that can broaden your reach for recruitment but that’s not their only value to your research. Are we communicating effectively from the beginning to end. Is the developing safety profile acceptable not just from a clinical or regulatory perspective but from a patients perspectives.
And finally, framing the results of a study from a consumer perspective can help with credibility in the patient population, and dissemination and implementation of the outcomes of the research.
And if you not yet convinced of what might seem subjective benefits that are hard to measure, the financial case for involvement is clear.

This US based research from a few years ago modelled the financial impact of consumer involvement throughout the clinical development phases and found significant financial benefit was gained through the avoidance protocol amendments, improved recruitment and retention, and ultimately in informing no-go decisions.
When research funding (public or commercial) is so scarce, we need to make sure every dollar spent wisely. While involving consumers may add an initial upfront cost to the budget, the ultimate savings in time and money as well as the outcome of having more relevant, recruitable, efficient, implemented research with less research waste has to make it worth it.
The statement is it currently stands lays out very nicely in the forward the fundamental principles and mindsets that I believe should carry into the future.

Consumers and the community offer unique and valuable insights as research is framed, conducted and translated, helping to ensure research quality and relevance.
As an extension to that, beyond the larger societal benefit, I believe it also helps provide focus to, humanise and improve the experience of the people participating in the research rather than treating them as a means to an end.
The statement also recognises that the form and degree of community involvement must be appropriate to the research activity and requires consumers and community members with differing backgrounds, interests and perspectives.
The values of shared understanding, respect and commitment, I believe should endure moving forward.
And yet, while this statement has been around for six years and supersedes statements that have been around for 20+ years, we know that the experiences of consumers in research are not always good nor the involvement meaningful.

At worst there is box ticking, being seen to be doing it, its dismissive of the experience or needs of consumers. There can be poor communication, a lack of respect for time or the financial impacts of involvement. It isn’t always inclusive. And when poorly recognised, power dynamics get in the way.
Meaningful involvement is collaborative, it’s a partnership, it’s having shared goals and working together to achieve those goals. It’s ensuring consumers feel valued and know the impact on their contributions, This is especially important when too often those contributions are expected to be made voluntarily.
The current vision is nice, but to my mind doesn’t ask enough of medical research itself. I think there is the opportunity to make health and medical research more accountable to the communities it serves and to the participants of that research.
Maybe something like - Health and medical research advances the knowledge health and well-being of all Australians in a way that is scientifically rigorous and transparent, relevant to and inclusive of the needs of a diverse Australia and respectful to those to participate and contribute to it.

By making health and medical research more accountable to the community, it is much harder to exclude consumer involvement as a necessity to the responsible conduct of research.
And to that end, while its useful to have a consumer involvement statement, I’d like to make sure we get away from thinking of consumer involvement as something extra bolted on or separate to the core business of research.
These are the principles of the current Australian code for the responsible conduct of research, which set out nicely the expectations.

As I read through each principle, it seems clear to me, knowing the potential impacts, how involving consumer is core to achieving many of these principle. However, there is really no reference in the descriptive text for each principle around consumer involvement.
As the consumer involvement statement evolves into a principles based document, I would encourage us to think about how it can be aligned and integrated with other recognised principles for the conduct of research so that consumer involvement becomes a core element (rather than a nice add on) to how we responsibly conduct research.
In wrapping up, there is no one way to involve consumers across a research project or with the research structures.
Early and consistent involvement and partnership, shared goals, shared responsibility for achieving those goals, that’s where we want to be. A principles based document is a way forward, but in my view will be redundant if it is not backed up with financial investment and accountability to the principles, to drive the change in culture needed and reap the benefits meaningful consumer involvement can bring and that have been talked about for decades now.
Thanks for allowing me to share these thoughts with you.
